March 2024
Process Optimization
An innovative process for simultaneously producing high-purity benzene and U.S.-grade gasoline from FCC C6 heartcut
Mobile Source Air Toxics (MSAT) II regulations restrict the average benzene level in gasoline sold in the U.S. to 0.62 vol%. In India and in several other countries, benzene in gasoline is presently restricted to 1 vol%. Fluid catalytic cracking (FCC) gasoline contributes to nearly 10%–20% of the gasoline pool in a typical refinery.
Mobile Source Air Toxics (MSAT) II regulations restrict the average benzene level in gasoline sold in the U.S. to 0.62 vol%. In India and in several other countries, benzene in gasoline is presently restricted to 1 vol%. Fluid catalytic cracking (FCC) gasoline contributes to nearly 10%–20% of the gasoline pool in a typical refinery. Full-range FCC gasoline contains around 15%–30% aromatics, with up to 2% benzene and 1,000 ppm–2,000 parts per million (ppm) sulfur. A narrow C6 heartcut fraction obtained from full-range gasoline contains between 11 wt%–19 wt% benzene and up to 150 ppm sulfur. In addition, if unprocessed, this fraction also contains reactive impurities such as oxygenates, metals, chlorides, sulfur and nitrogen compounds, diolefins and organic peroxides. Therefore, the removal of benzene from this heartcut and its subsequent blending in the gasoline pool would help refiners meet the above levels of benzene in the overall gasoline pool. However, the complexity of this feedstock has hindered the development of an economic and reliable benzene recovery process in the industry. Hydroprocessing routes for benzene reduction result in olefin saturation, which lowers the octane rating of the final product.
Reliance Industries Ltd. (RIL) operates two 200,000-bpd FCC units (FCCUs) at its world-class refineries and petrochemical complex at Jamnagar, India. The gasoline from these two FCCUs is a major contributor of benzene in the gasoline pool. Therefore, it seemed imperative to remove benzene from this stream to adhere to the desired benzene limit in the gasoline pool. To address the challenge of the unavailability of a proven commercial process, the co-authors’ organizationsa collaborated to jointly develop a novel technology for processing the C6 heartcut of FCC gasoline—based on the principles of extractive distillation—without the requirement for any pre-processing step to saturate diolefins and reduce reactive impurities (e.g., chlorides, peroxides). This technology serves the dual role of not only producing U.S.-grade gasoline but also recovering high-purity benzene.
The unique and novel aspects of the technology are:
- It is based on a thermally and chemically stable and tunable solvent system designed to handle reactive impurities in FCC gasoline.
- The innovative and proprietary process configuration minimizes solvent loss and utility requirements, and maximizes purity and yield of products.
- It does not require any prior hydrogenation step to saturate diolefins, which makes the process a simple, energy efficient and low-cost operation.
- The process guarantees the benzene content of the raffinate (gasoline) below 0.1%, with benzene recovery exceeding 99%, while producing extract with more than 98% aromatics. Moreover, the octane of gasoline is preserved since olefins in the feed are retained.
Solvent selection
Several extractive distillation-based technologies are available from various licensors for benzene, toluene and xylene (BTX) recovery from reformate and hydrogenated pyrolysis gasoline. Several commercial units are operating around the world on these feedstocks. However, both of these feedstocks are almost void of any olefins and reactive impurities. There is no instance of an extractive distillation unit operating anywhere in the world that is processing an impurity-laden FCC gasoline heartcut fraction for the dual purpose of producing benzene-lean gasoline and high-purity benzene.
The primary challenge in processing impurities-laden FCC heartcut gasoline is the presence of diolefins and peroxides in the feedstock, and the tendency of cracked gasoline to react with oxygen and form peroxides. Organic peroxides interact with diolefins (especially conjugated diolefins) in the feed, initiating polymerization reactions due to auto-oxidation. The presence of diolefins and olefins together form more peroxidic compounds than when present individually in the same concentration. This reactive mixture can cause degradation of the solvent, particularly at the high temperatures encountered in the extractive distillation column (EDC) and solvent recovery column (SRC).
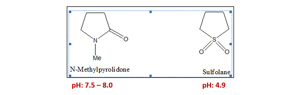
Alkenes are also known to undergo polymerization at high reaction temperatures in polar mediums under acidic conditions. Instances of such polymerization—especially when conjugated diolefins are present—have been reported many times. For the selection of the optimum solvent, various solvent systems were tested for their oxidative and thermal stability under temperature conditions normally encountered in extractive distillation. The conventional solvent systems based on sulfolane used in extractive distillation are highly unstable at higher temperatures due to the presence of a highly polar S=O bond (FIG. 1), in view of its lower bond strength. This accelerates polymerization, resulting in solvent degradation at higher temperatures. The innovative nitrogen-based solvent system of N-methyl-2-pyrrolidone (NMP) has a C=O bond, which is very stable compared to S=O since the polarity and acidic character of the C=O bond is much lower and does not initiate the polymerization of diolefins.
A proprietary co-solvent was identified and mixed at an optimum proportion with NMP to maximize the selectivity and capacity of the overall solvent system, as well as to minimize the polymerization and degradation of the solvent. The extent of the co-solvent can be tuned as per requirements, which is also an added advantage with this solvent system.
Rigorous testing of this solvent system (NMP with a co-solvent) was carried out to establish the extent of polymerization and its impact, if any, on the corrosion of carbon-steel metallurgy commonly used in the construction of such plants. This was carried out to eliminate the possibility of reducing the efficacy of the separation process due to degraded solvent and to alleviate corrosion in equipment, especially in high-temperature zones like tower bottoms and heat exchangers.
Therefore, choosing the right solvent combination—a combination that is stable for this hydrocarbon feed at the operating conditions and is suitable to achieve the objective of the technology—establishes the technical viability of the process.
Experimental verification and further studies for process optimization
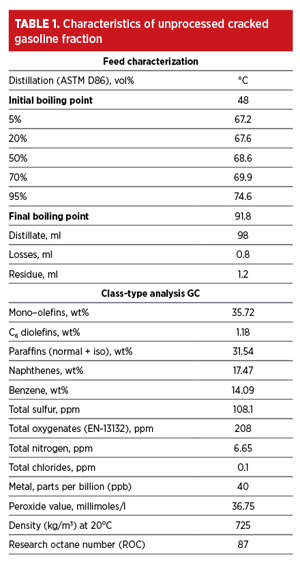
The characteristics of FCC heartcut gasoline used in the experimental studies are provided in TABLE 1. As shown in TABLE 1, the aromatic content of the feed was low (~14 wt%), whereas the olefinic and naphthenic contents were relatively high at 37 wt% and 17 wt%, respectively. This is quite the opposite to conventional feedstocks—such as reformate and pyrolysis gasoline (widely used for aromatics production)—where the aromatic content is more than 60% with relatively low amounts of non-aromatics, particularly naphthenes and olefins. The production of pure aromatics from such low aromatic feedstocks presents a formidable challenge in view of the relative selectivity and solubility of olefins, naphthenes and aromatics in any solvent.
In view of the above, a series of carefully planned extractive distillation experiments was carried out: first, to experimentally verify the efficacy and stability of the solvent system to meet the project objectives, and, second, to generate data to establish the effect of column operating variables on the benzene content in the raffinate and the purity of the aromatics in the extract. These runs were carried out on a 25-mm inner-diameter laboratory columnb. Utmost care was taken to achieve good (within 5%) material balance around the process.
The effects of various column operating variables—such as reboiler temperature, reflux rate, solvent-to-feed (S/F) ratio and column pressure—were studied to establish the optimal set of operating parameters.
Note: It was rewarding that, in these columnb runs—without exception—the project operating objectives were completely met: the aromatics purity in the extract was more than 95%, and benzene recovery exceeded 99%, with benzene levels in the raffinate < 0.2%. In addition, all sulfur compounds—such as thiophenes and benzothiophenes—were extracted into the extract, along with the aromatics, thus providing a benzene- and sulfur-free raffinate, a premium gasoline blendstock.
Modeling of vapor-liquid equilibrium (VLE) development of rigorous simulation model
A typical extractive distillation process uses a separation solvent that interacts non-ideally with the components of the mixture, thereby causing their relative volatilities to change. For the authors’ system, this results in higher volatility of the non-aromatics so that it separates out as the top product. The bottom product consists of a mixture of the solvent and aromatics, which is then separated easily in an SRC. For non-ideal systems, the relative volatility between two components is given in Eq. 1:
where the components A and B represent the non-aromatics and aromatics, respectively.
For a particular component, ƒi is the fugacity coefficient of the vapor phase (estimated from equations of state), ρoi is the vapor pressure, while ϒi is the liquid phase activity coefficient generally estimated from solution theories. The authors preferred to use the non-random two-liquid (NRTL) mixing model1 provided by Eq. 2:
where:
The key in modeling an extractive distillation process is to determine the liquid phase activity coefficients, which, in turn, requires the accurate estimation of the NRTL interaction coefficients (τi j and αi j).
The first step is to identify the key components in the hydrocarbon feedstock by utilizing detailed hydrocarbon analysis (DHA). Although the FCC heart-cut gasoline is a narrow 60–90 cut, the DHA revealed several hydrocarbon components with different polarities and boiling points.
The next step is to estimate the NRTL interaction coefficients (τi j and αi j ). These were estimated from the actual VLE/liquid-liquid equilibrium (LLE) data generated in the laboratory. Several multi-component VLE datasets were also generated using the actual feedstock; the NRTL parameters were then fine-tuned by matching this multi-component VLE data.
Process simulation and optimization
A commercial flowsheet simulator was used to simulate the entire process. The model included the EDC and the SRC embedded in the entire flowsheet of the process. This model was then utilized to scale up to commercial design capacity and to optimize all unit operations simultaneously in the flowsheet. The scale-up from a 25-mm-diameter column to a commercial EDC—approximately 200 times larger in size—was carried out without recourse to an intermediate scale pilot or demo plant. This approach expedited the technology development from an idea to a commercial scale in < 18 mos. Note: This fast-tracking process was possible due to the development of an accurate and rigorous simulation model and its validation with a large number of well-planned experimental runs on the laboratory column.
Out-of-the-box process configuration
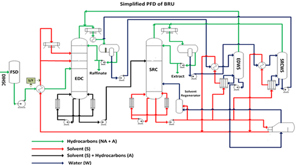
The technology offers an innovative process flowsheet (FIG. 2). This flowsheet serves the dual purpose of minimizing solvent loss and maximizing product recoveries. In this process, the solvent system and preheated unprocessed cracked gasoline fraction (FCC C6 heartcut) are treated in an EDC, where the untreated cracked gasoline fraction enters the EDC at the middle section, and solvent enters the column at the top section below the reflux entry point. The benzene lean gasoline (raffinate) product is recovered from the overhead of the EDC, while the benzene loaded solvent exits from the bottom of the EDC. Solvent from aromatic-rich hydrocarbon is separated from the SRC. The hydrocarbon-free solvent is obtained from the SRC bottom, while aromatic hydrocarbons are obtained from the SRC overhead. Adequate reflux is provided in both the EDC and the SRC to minimize solvent carryover into the overhead line. Also, a solvent regeneration column is provided to remove any polymerized/degraded material accumulated in the circulating solvent over a long operating period.
Two water stripper units—namely an extractive distillation water stripper (EDWS) and a SRC water stripper (SRCWS)—are provided with no additional utility costs. Both the strippers run on the heat provided by the hot circulating solvent from the SRC bottom. The EDWS helps to recover any dissolved non-aromatic hydrocarbons in the EDC overhead boot water. Conversely, the SRCWS helps to recover any dissolved aromatic hydrocarbons in the SRC overhead boot water. The two water stripper units do not allow the boot water to escape from the unit. Generally, as compared to hydrocarbons, this water has a relatively higher quantity of solvent and would have otherwise resulted in solvent losses from the system.
A comprehensive pinch analysis was carried out to identify various hot and cold streams. While the process has only one hot stream (namely the circulating solvent), it has several cold streams: the reboiler duties of all columns and feed preheating of all columns, including feed vaporization, as well as vaporizing the water to produce a stripping stream for the SRC. Therefore, the circulating solvent stream is intricately run within the entire process plant with branching and recombining so that it can provide heat in the stripper reboilers and stripper feed preheaters, the process vapor generator and the EDC feed preheater. This heat integration reduced the net energy consumption of the process, thus resulting in reduced Scope 1 emissions.
This simple and out-of-the-box innovative process configuration is the first of its kind. It comprises a highly efficient system of interconnected process loops, which minimizes product and solvent losses, and ensures high-purity product streams with minimum utility requirements. The annual solvent system makeup is very low, even when compared to existing traditional extractive distillation units.
Process design for reliability and sustainability
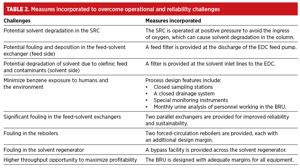
The commercial benzene recovery unit (BRU) at RIL’s Jamnagar refinery has been designed with state-of-the-art engineering principles to make the process highly reliable and sustainable. TABLE 2 provides the operating flexibility and high-reliability measures incorporated against the possible challenges. The solvent system selected is extremely stable, both thermally and oxidatively. Nevertheless, provisions were made in case the solvent experienced any degradation.
Innovative features of the technology
The technology has several benefits:
- The process does not require any prior hydrogenation step to saturate the diolefins, making the process simple, energy-efficient and cost-effective.
- All unit columns (EDC, SRC and strippers) operate above atmospheric pressure (no vacuum), which eliminates any possibility of oxygen ingress, thus eliminating the possibility of chemical degradation of the circulating solvent.
- The solvent selectively removes sulfur compounds from the feed and provides a nearly sulfur-free (< 5 ppm) dearomatized gasoline. The sulfur removal from gasoline (raffinate) without hydrogenation ensures no octane loss because of olefin saturation. However, there is a small octane reduction due to benzene (RON-104) removal from the raffinate going to the gasoline pool.
- The technology simultaneously provides the production of low-benzene gasoline and the recovery of high-purity benzene; the recovery and purity of benzene is very high (> 99 wt% and > 97 wt%, respectively). The aromatics content in the raffinate is consistently < 0.2%.
- The solvent/co-solvent combination is thermally and oxidatively stable throughout the operation and can process the hydrocarbon feedstock, even in the presence of highly reactive impurities, including organic peroxides.
Technology commercialization
A 700,000-tpy commercial unit—based on the innovative technology—was built at RIL’s Jamnagar refinery. The unit was successfully commissioned in May 2016. FIG. 3 shows the unit at the RIL site. The unit’s commissioning was flawless and extremely smooth. No teething problems were encountered during commissioning, and on-spec product was routed to the product tanks within 48 hr of the feed cut-in.
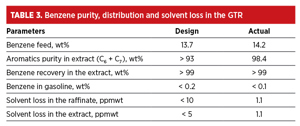
Once the steady-state operation was achieved, the plant throughput was brought up to the design capacity. It was decided to carry out a guarantee test run (GTR) and a post-audit to establish the performance of the unit vis-à-vis design. The design and actual aromatics purity and distribution, product yield and solvent loss are compared in TABLE 3.
It can be seen from TABLE 3 that, even at a higher benzene feed content, the unit produced on-spec gasoline with almost no solvent loss through the products. The aromatics recovery (> 99%) and purity (98.4%) were better than in the design case, as well.
Based on the unit’s performance, the technology was declared a commercial success. Since commercial startup, the unit has been in operation consistently and has even ramped up to a throughput of 150% of design. To date, after more than 50,000 hr of operation, the unit has not undergone any turnaround, and hardly any solvent makeup is required, since the solvent degradation is essentially zero. No reliability issues have been encountered in operating the unit since startup.
With the commercialization of this technology, RIL’s Jamnagar refinery is the only refinery in the world to have the capability to produce high-purity benzene from FCC gasoline heartcut. RIL and CSIR IIP are in advanced stages of discussion with major technology licensors to license the technology worldwide.
This technology empowers refiners to achieve their goal toward a cleaner and safer environment, and to enable the transition toward crude oil-to-chemicals (COTC). The absence of such a unit in a refinery configuration would likely lead to the downgrade of FCC gasoline to naphtha or for it to be hydrotreated for blending in the gasoline pool with benzene loss—effectively, a financial penalty either way.
Greenhouse gas (GHG) footprint
Three kinds of GHG emissions (namely Scopes 1, 2 and 3) are attributed to plant operations. While Scope 1 and Scope 2 emissions are attributed to their own operation, Scope 3 emissions pertain to the wider downstream value chain. It is difficult and challenging to compare Scope 1 and Scope 2 GHG emissions of the benzene recovery technology with other available technologies, since these follow different routes to reduce benzene—by either benzene saturation or benzene alkylation. Conversely, since the benzene recovery technology recovers benzene as a byproduct from the FCC C6 heartcut, unlike hydrotreating and benzene alkylation technologies, it reduces the overall fuel basket by approximately 12%–18%, resulting in a reduction in Scope 3 emissions. As a byproduct, the high-purity benzene produced from this technology has a minimal GHG footprint.
Takeaway
This first-of-its-kind technology—the BRU—allows refiners to recover benzene, a primary building block for petrochemicals, and, simultaneously, to produce benzene-lean gasoline from FCC heartcut naphtha. The technology has been a commercial success at RIL’s Jamnagar refinery. The first-ever unit built and commissioned in May 2016 has been in operation for 7 yr without a turnaround. This technology has the potential to be a key unit in the global efforts to convert crude to chemicals, and to achieve targets of decarbonization in the foreseeable future. HP
NOTES
a RIL and CSIR–Indian Institute of Petroleum
b Oldershaw distillation column
LITERATURE CITED
1 Renon, H. and J. M. Prausnitz, “Local compositions in thermodynamic excess functions for liquid mixtures,” AIChE, 1968.




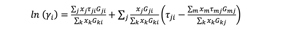
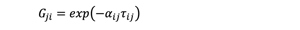









Comments