March 2026
Special Focus—Petrochemical Technologies
Cutting-edge catalytic technologies deployment in a modern aromatics complex
Global demand for paraxylene is supported by long-term fundamental growth of packaging, bottling and textile products, despite recent softness due to significant capacity increases as a result of newly built crude-to-chemicals complexes. Focusing on China, there have been visible trade gaps between domestic paraxylene (PX) production and purified terephthalic acid (PTA) plants consumption before reaching self-sufficiency. In addition, older plants producing PX with poor scale economics and low energy efficiency are likely to be rationalized and replaced by newer and larger manufacturing facilities that employ state-of-the-art technologies. Recognizing this continuous market need, CNOOC Huizhou Petrochemical Co. Ltd. (CHPCL) has expanded its production capabilities by commissioning a new aromatics complex in Daya Bay, China. This facility is designed to produce 1.5 MMtpy of PX using state-of-the-art technologies from the author’s (ExxonMobil) and co-author's (Axens) companies.
The new aromatics complex is part of the CNOOC Huizhou Petrochemical Product Structure Optimization and Quality Upgrading Project, a key initiative by Guangdong Province and CNOOC, and is one of the most recent crude-to-chemical facilities in the world. It includes major processes such as catalytic reforming, hydrocracking, mogas etherification and aromatics recovery, along with public utilities and auxiliary facilities. This project aims to balance the materials of the entire plant, enrich the product structure, and accelerate transformation and upgrading while maintaining the existing crude oil processing capability in the scale of 22 MMtpy. The aromatics complex was licensed and began construction in 2018 and successfully commissioned in October 2023. The complex has successfully passed performance tests at its design capacity. The annual production value is 15 billion (B) RMB (~$2 B).
The new aromatics complex employs the co-author’s company’s technology suitea, which includes several advanced processes for the efficient production of aromatics. FIG. 1 shows the overall schematics of the complex. As illustrated, in addition to the typical processing of reformate streams from the CNOOC refinery, the scheme is tailored to upgrade lower value toluene and pyrolysis gasoline cuts from existing assets to value added products such as benzene and PX, together with a side production of mixed xylenes, either to feed CNOOC’s existing PX plant or for export as market grade xylenes for PX production.
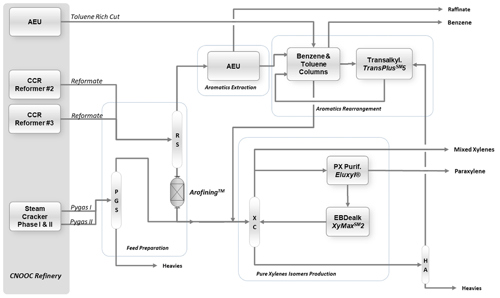
FIG. 1. Schematics of CHPCL aromatics complex process blocks.
The key technologies integrated into the complex are highlighted as follows:
- A heavy aromatics transalkylation processb, developed by the author’s company, which converts C9+ aromatics and toluene into higher-value mixed xylenes and high-purity benzene. The process features a high-activity zeolite catalyst, offering superior yields, longer cycle length and lower operating costs.
- A vapor-phase xylenes isomerization technologyc, also from the author’s company, uses its latest proprietary zeolite catalyst that provides ultra-low xylene loss, high ethylbenzene (EB) conversion, increased weight hourly space velocity (WHSV) and an expanded temperature window, enhancing the overall energy efficiency of the xylene isomerization process by reducing the xylene loop traffic.
- This paraxylene purification technologyd, developed by the co-author’s company, employs a state-of-the-art simulated counter-current adsorption concept using the newest generation of molecular sieves in a cost-effective single adsorbent tower design. This technologyd enables the production of very high-purity PX with high recovery rates, making it a benchmark technology for PX purification.
- This selective hydrogenation technologye, also from the co-author’s company, is designed to remove unsaturated compounds from aromatic-rich feeds without aromatics ring loss. The olefins-free aromatics feed helps to achieve longer cycle operations of downstream catalysts, while the absence of aromatics rings losses combined with the conversion of styrenics to saturated aromatics enables PX and benzene production gain.
Challenges and solutions. Building a new PX complex involves overcoming numerous challenges related to technology, logistics, workforce management, government policies compliance and environmental impact—specifically solid waste. The impacts of the COVID-19 pandemic also created additional, unprecedented difficulties. The key challenge was to deliver a new, highly integrated aromatics complex quickly to meet market demands in the very competitive Chinese market, where efficiency is valued.
First and foremost, implementing innovative technical solutions and best practices is vital to addressing these difficulties. CHPCL has been at the forefront of adopting advanced refining and petrochemical technologies to enhance its production capabilities to drive down manufacturing costs. Since 2009, CHPCL has been satisfactorily operating a complex using the co-author’s company’s technology suitea with a production rate exceeding 1.3 MMtpy. In the technology licensor selection stage for this new complex, CHPCL thoroughly evaluated proposals from multiple technology providers and eventually determined that the co-author’s company’s design provided the best solution with state-of-the-art technologies and optimized integration.
As the lead licensor, the co-author’s company integrated its own paraxylene purification processes with its technology suitea alliance partner, ExxonMobil Catalysts and Licensing LLC’s (EMCL’s) latest catalytic conversion technology advancements into CHPCL’s petrochemical complex in Daya Bay, China. The goal of this integration, as part of a broader expansion project, was to increase the existing production capacity of PX to 3 MMtpy. The state-of-the-art aromatics complex not only boosts production capacity but also enhances energy efficiency and reduces operational costs due to the superior aromatics yields provided by the advanced reaction/adsorption technologies and optimized heat integration.
TECHNOLOGY PERFORMANCE
The author’s company’s vapor-phase xylenes isomerization technologyc. This process is the state-of-the-art xylene isomerization technology that uses the author’s company’s proprietary catalyst, which has demonstrated unmatched stability over > 100 yrs of cumulated commercial experience since commercialization. This proprietary technologyc process has commercial applications in both axial and radial flow reactor configurations at more than a dozen PX manufacturing plants worldwide, including multiple customers that converted the other xylenes isomerization processes to this vapor-phase xylenes isomerization technologyc.
In this process, a para-depleted xylene feed is isomerized to an equilibrium mixture of xylenes for further recycle to increase PX recovery in the xylene loop. The EB in the feed is converted via de-alkylation to benzene and ethylene byproducts. The ethylene is further saturated to a liquefied petroleum gas (LPG) component or cracker feed-ethane. This chemistry is illustrated in FIG. 2.
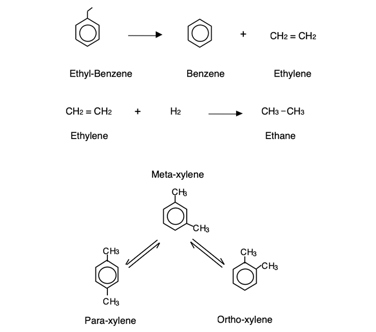
FIG. 2. Xylenes isomerization primary reactions.
Several side reactions may occur in addition to the main reactions described above. Examples of these undesired reaction chemistries are shown in FIG. 3. Transalkylation reactions, such as the reaction of EB with xylenes, form benzene, toluene and C9+ aromatics. Ring loss can occur through the hydrocracking of aromatics to form light gases, and condensation of aromatic rings can lead to coking, which in turn decreases the effectiveness of the catalyst.
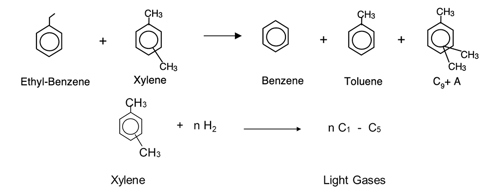
FIG. 3. Examples of xylenes isomerization secondary reactions.
It is desirable in a high energy efficiency complex that the xylene loop recycle traffic is minimized as much as possible, since > 75% of the total feed to the PX separation (e.g., the co-author’s company’s paraxylene purification technologyd) is recycled, which results in large energy consumption due to multiple phase changes of the processed streams.1 A key driver to high yield is to push for higher EB conversion (EBC) to lower the xylenes loop traffic. However, driving higher EBCs in many processes also drives side rate reactions that lead to higher xylene loss, and the process will require significantly more raw material (naphtha, reformate, etc.) to maintain fixed PX production rates.
Although xylenes lost to toluene and C9+ aromatics can be reclaimed by an aromatics transalkylation process, this substantially increases the energy consumption to complete such a large loop recovery, which is counterproductive to an energy efficient process. Therefore, an ideal complex design would set high EBCs while achieving low xylene loss, which is often extremely challenging to the catalyst technology providers since high EBC at a certain WHSV requires high reactor temperature that intensifies undesirable side reactions. The vapor-phase xylenes isomerization processc, as part of the technology suitea alliance, operates at high EBC while maintaining low side rate reactions per pass, which reduces xylene loop traffic leading to higher xylene concentration and increased PX recovery.
The innovative solution provided by the vapor-phase xylenes isomerization processc attributes to the proprietary catalyst manufactured using the author’s company’s legacy zeolite shape selectivation expertise. The unique design of the catalyst tailors zeolite pores to allow preferential diffusion of the linear structured EB molecule with smaller kinetic diameter (5.8 Å) over larger molecules. The larger ortho-xylene (OX) and meta-xylene (MX) molecules (6.8 Å) will bypass the first bed to minimize side reactions. This enables the use of zeolite with a low silicon/aluminum (Si/Al) ratio, which generates high acidity that enhances EB conversion, as shown in FIG. 4.
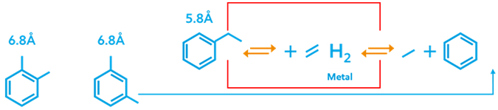
FIG. 4. Zeolite catalyst design for xylenes isomerization.
Moreover, the external acidity is tailored to reduce any reaction so that xylenes isomers bypass the first bed chemically to avoid side reactions despite the high acidity in internal pores.
The author’s company’s vapor-phase xylenes isomerization technologyc operates at lower temperature and high ethylbenzene conversion, provides very low xylene loss with negligible ring loss, and delivers steady yields throughout the entire long cycle. At CHPCL’s new complex, this technologyc has unsurprisingly been repeating its legacy performance by delivering stable operation, decreased energy consumption and improved yields. TABLE 1 shows the results of key performance parameters that meet or exceed the design benchmark, with ultra-low xylene loss, high EB conversion, high PX selectivity and low chemical hydrogen (H2) consumption.

In subsequent continuous monitoring provided by the author’s company, the vapor-phase xylenes isomerization processc has been providing very stable and excellent performance, shown in FIG. 5. Also shown in FIG. 5 is that the xylene loss reduces over time as the catalyst continues to selectivate towards preferred products.
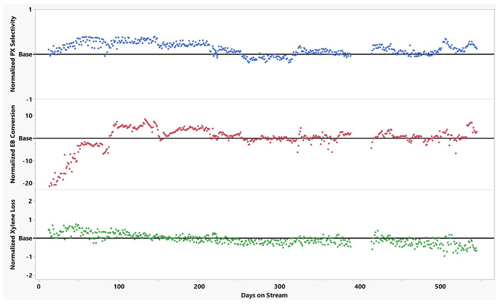
FIG. 5. The author’s company’s vapor-phase xylenes isomerization technologyc unit key performance data.
The author’s company’s heavy aromatics transalkylation processb. This process is the next-generation aromatics transalkylation technology that is noted for its improved ability to upgrade C9+ aromatics and toluene into higher-value mixed xylenes and high-purity benzene products. The process has shown substantial improvement over its predecessors, including higher yields, longer catalyst cycles and reduced operating costs.
The aromatics transalkylation processb is based on a proprietary high-activity zeolite catalyst. From discovery to deployment of the aromatics transalkylation catalyst, the author’s company has invested significant research and development (R&D) effort to drive continuous improvement of the catalyst performance. To meet the industry’s increasing performance standards, a fundamental understanding of process chemistry is the key for success. As illustrated in FIG. 6, alkylbenzene dealkylation and transalkylation are desired reactions, while alkylation and ring cracking or saturation are undesired.
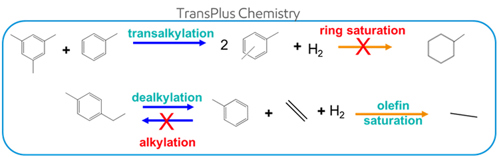
FIG. 6. The chemistry of the author’s company’s heavy aromatics transalkylation processb.
In the catalyst design, balancing metal functionality to promote olefin/ring saturation with zeolite acidity that catalyzes dealkylation and transalkylation is a challenge. This is done through the optimization of the metal type, zeolite synthesis to control acidity, and careful impregnation to ensure acid-metal intimacy. The author’s company has > 50 yrs of experience in inventing and producing catalysts that are optimized for performance.
As an example of catalyst design, design of experiment and high throughput testing tools were used to accelerate formulation optimization. Several factors were considered in the design, including but not limited to zeolite type(s), metal type, metal loading, binder content, activation and calcination. An optimized catalyst formation with a zeolite framework of Mordenite (MOR) and Mobil-type five (MFI) family active materials synergistically integrated in the structure design and manufacturing processes was found to maximize xylene yield and produce high-purity benzene. Additionally, the metal type and load amount were studied to promote in-situ olefins saturation to minimize coke precursor formation and prevent catalyst aging, while minimizing ring saturation to enable high benzene purity. The method of metal binding to the zeolite and its support were also studied. It was found that simple incipient wetness impregnation often led to metal being concentrated on the rim of the catalyst, which in turn results in poor performance. An optimized metal binding procedure, conversely, provides substantial improvement in xylenes yield, as shown in FIG. 7.2
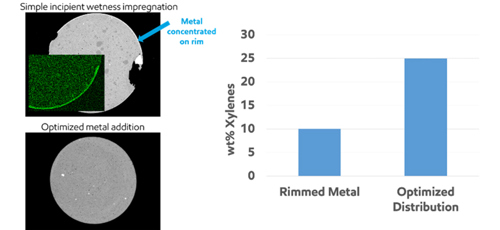
FIG. 7. Metal-zeolite bounding optimization.
The multi-function catalyst effectively optimizes the chemistry of alkylbenzene dealkylation, disproportionation/transalkylation and non-aromatics cracking to obtain maximum benzene and C8s yields. The advanced catalyst is designed to achieve high de-ethylation rate, near equilibrium xylenes yield and high benzene purity with low metal content. The main advantage of the author’s company’s heavy aromatics transalkylation processb catalyst is the ability to operate at high throughputs and conversion levels, and to process high C9+-containing feeds while maintaining low aging rates. The catalyst for the heavy aromatics transalkylation processb process improves benzene purity, resulting in reduced stabilizer side draw flow and decreased extraction unit energy usage. Key technical features include:
- Broader operating window:
- Operates within an expanded temperature window, providing greater flexibility and low energy use.
- Operates at a low H2:HC ratio, avoiding large recycle gas compressor investment and subsequent operating cost.
- High toluene + C9+ conversion: The proprietary transalkylation processb achieves higher total conversion (toluene and C9+ aromatics) per pass, reducing recycle rates and operating costs.
- High throughput: The transalkylation processb operates at a high WHSV, allowing high reactor feed rate or reduced catalyst load size. For grassroots units like the CHPCL transalkylation processb, this translates to a smaller unit size for reduced capital expenditure.
- Enhanced selectivity: The transalkylation processb offers superior selectivity to xylenes and benzene products, which enhances complex production efficiency
- Minimizing aromatics ring loss: Reduced light gas yield minimizing liquid chemical value downgrade to LPG and low chemical H2 consumption.
- Broad feedstock flexibility: Capable of handling fresh feeds with compositions ranging from very light to 100% C9+ aromatics; and allows co-processing benzene.
- Long cycle length and in-situ regeneration: The catalyst for the author’s company’s heavy aromatics transalkylation processb aging rate has been greatly reduced compared to its prior version, which meets or exceeds the plant turnaround cycle requirement. The catalyst is also fully regenerable in-situ, allowing for multiple cycles operation and extended total life.
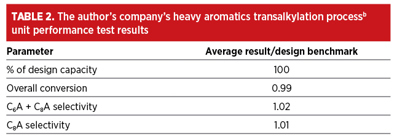
In a performance test at a design complex PX production capacity, the heavy aromatics transalkylation processb demonstrated excellent performance, as expected. TABLE 2 shows the results of key performance parameters that meet or exceed the design benchmark, including high total toluene/C9+ conversion as well as high benzene and xylenes yields.
In subsequent continuous catalyst monitoring provided by the author’s company, the transalkylation processb unit continues to provide stable and excellent performance, shown in FIG. 8.
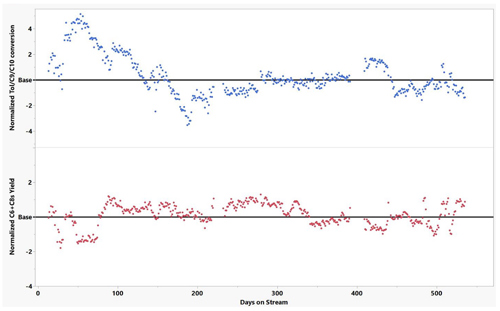
FIG. 8. The author’s company’s heavy aromatics transalkylation processb unit performance.
The co-author’s company’s selective hydrogenation technologye. High-severity continuous catalytic reforming (CCR) units employed to maximize aromatics production are operated under conditions that inevitably promote the formation of unsaturated compounds. These compounds are detrimental to the reliability of downstream aromatics transformation processes and may jeopardize the attainment of final products specification.
The co-author’s company’s selective hydrogenation technologye is a selective hydrogenation process for olefins, diolefins and alkenyl aromatics conversion to paraffins and alkyl aromatics in an aromatics stream. It stands out from other olefins removal processes due to its unique selectivity.
Yielding additional aromatics. Aromatics retention is a key parameter for aromatic complex profitability. It is essential to preserve native aromatics from reformate along each transformation step of the aromatics complex, and especially during the olefins removal step.
- Historical clay treaters solution for olefins removal represents direct aromatics product loss: Acid alkylation of reformate olefins with aromatic molecules occurs on clays, thereby producing heavier aromatics compounds that are purged out of the aromatics complex by fractionation as a low-value heavy stream (typically fuel oil). Therefore, clay treatment inherently reduces PX and benzene production in the effort to remove undesirable olefins.
- The selective hydrogenation solutione directly improves the profitability of the aromatics complex: As opposed to direct aromatic loss during clay treatment, the selective hydrogenation processe eliminates olefins, with a net gain in aromatics yields. The selective hydrogenation processe hydrogenates olefins present in the lighter C7- reformate fraction in their corresponding saturated paraffins with virtually no aromatics loss, which is a first benefit over clay treatment.
In addition, styrenics can account for > 95% of the unsaturated compounds to be removed in a C8+ heavier reformate. With the co-author’s company’s selective hydrogenation processe, the hydrogenation of these undesirable styrenics produces additional valuable aromatics, essentially doubling the benefit noted above.
Environmental considerations. Spent clay from clay treaters is a yearly waste product from an aromatics plant that represents a compounding waste over the years, as illustrated in FIG. 9. Typically, spent clay can be a significant waste stream of an aromatics complex. It represents up to 80% of an aromatics complex’s total solid waste.
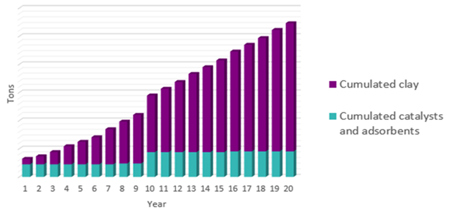
FIG. 9. Solid wastes in an aromatics complex.
With selective hydrogenation, the deactivation of clay is eliminated or drastically reduced, resulting in significant reductions of both solid waste generation and disposal costs. Associated wastewater generated from activation of clay and steam stripping before clay unloading is also considerably reduced with the selective hydrogenation technologye.3
Selectivity. When processing streams primarily containing aromatic compounds, having aromatics hydrogenation side reactions developing during hydrogenation catalyst cycle and leading to an increasing loss of aromatics may impact plant economics.
The co-author’s company’s selective hydrogenation catalystf stands out from the market thanks to its superior aromatics retention. Its tailored metal function selectivity hydrogenates diolefins, olefins and alkenyl aromatics at a very low aromatic loss [100 parts per million (ppm)]. Other less selective catalysts formulations may feature aromatics loss in the range of 1% or higher, and these may further increase along catalyst cycle while the temperature is increased to compensate for activity loss. The cumulative discounted cash flow (CDCF) graph (FIG. 10) illustrates the sensitivity of various catalyst performance on the plant economics.
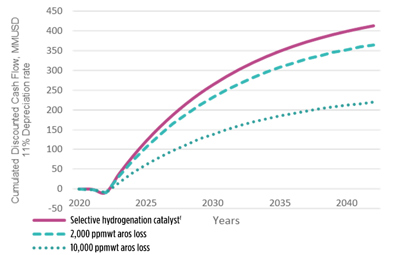
FIG. 10. Cumulative impact of aromatics loss on plant economics.
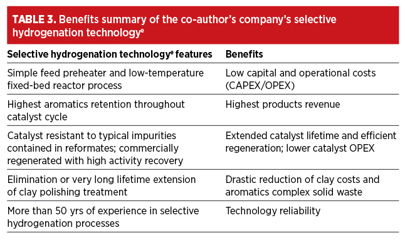
A benefits summary is listed in TABLE 3.
Complex integration. The CHPCL complex features the co-author’s company’s high energy efficiency design, which focuses on reducing energy consumption and increasing product yields. This design incorporates advanced heat integration techniques, optimized process flows and energy-efficient equipment, resulting in a highly efficient operation. Integrating an aromatics complex with advanced transalkylation, xylene isomerization and PX purification processes can significantly impact production costs in several ways:
- Improved yield and selectivity: Advanced catalysts and optimized process conditions in transalkylation and isomerization units increase the yield. Higher yields mean more product from the same amount of feedstock, reducing raw material costs per unit of product.
- OPEX reduction: By optimizing process configurations and technology selection, integrated complexes can reduce operational costs. This includes lower maintenance costs due to more efficient and reliable equipment.
- Reduced byproduct handling: Integration minimizes the production of unwanted byproducts, reducing handling and disposal costs.
- Energy efficiency: Advanced heat integration utilizes waste heat recovery systems to minimize energy consumption and improve overall process efficiency.
Overall, the integration of these advanced technologies has resulted in significant operational efficiencies. The author’s company’s heavy aromatics transalkylation processb allows for high conversion rates of heavy aromatics, reducing the need for recycling and lowering overall operating costs. The author’s company’s vapor-phase xylenes isomerization technologyc technology ensures optimal isomerization of xylenes, maximizing the production of para-xylene while minimizing energy consumption. The co-author’s company’s selective hydrogenation technologye further reduces the cost by minimizing the amount of aromatic loss endured to remove olefins. Finally, the other components not discussed in detail—such as the co-author’s company’s single adsorber technologyd that provides high-purity PX with excellent recovery rates—further enhance the overall efficiency of the complex.
Strategic significance for CHPCL. The successful commissioning of this new aromatics complex positions Huizhou Petrochemical as a leading producer of PX in the region. The facility's advanced design and excellent operation not only improve CHPCL’s resilience and competitiveness in the market, but also contribute to its broader goal of leading in energy efficiency and reduced solid waste. The adoption of new catalytic technologies by CHPCL exemplifies the company's commitment to leveraging advanced technologies for efficient petrochemical production of chemical intermediates. The use of the technologies detailed above has resulted in a highly efficient and competitive facility.
NOTES
a Axens’ ParamaX® Technology Suite
b ExxonMobil’s TransPlus℠ 5
c ExxonMobil’s XyMax℠-2
d Axens’ Eluxyl®
e Axens’ Arofining™
f Axens’ LD 267R Arofining® catalyst
LITERATURE CITED
1 Sakaguchi, T. and M. Molinier, “Maximizing existing xylene loop profitability,” Hydrocarbon Processing, January 2019.
2 Elia, C., et al., “From discovery to deployment of an aromatic transalkylation catalyst,” AIChE Annual Meeting, Boston, Massachusetts (U.S.), 2021.
3 Claire, F., A. Cotte and M. Molinier, “Enhance aromatics production with concurrent reduction of environmental footprint,” Hydrocarbon Processing, Special Focus, ME-TECH, Dubai, UAE, 2024.
The Authors
Related Articles
- Innovative in-situ batchwise/continuous regeneration scheme for high coke CCR reforming catalyst at the Saudi Aramco Jazan Refinery Complex
- Enhance plant performance and sustainability through activated carbon purification in gas sweetening: A Case Study
- Quantifying Europe’s steam cracking closures and implications for the polyethylene market








Comments